Seznamy 134+ Percentage Atom Economy Equation Výborně
Seznamy 134+ Percentage Atom Economy Equation Výborně. The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):
Prezentováno Quiz Worksheet Percentage Atom Economy Of A Reaction Study Com
The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.Percentage yield is calculated from the mass of reactants and the mass of products.
Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):.. Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation:. Percentage yield is calculated from the mass of reactants and the mass of products.

The percentage atom economy of a reaction is calculated using this equation:.. .. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product... The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Percentage yield is calculated from the mass of reactants and the mass of products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. Percentage yield is calculated from the mass of reactants and the mass of products.

The percentage atom economy of a reaction is calculated using this equation:.. Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):
The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):. Percentage yield is calculated from the mass of reactants and the mass of products.

Percentage yield is calculated from the mass of reactants and the mass of products... The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product... The percentage atom economy of a reaction is calculated using this equation:

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

Percentage yield is calculated from the mass of reactants and the mass of products. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy ….. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …
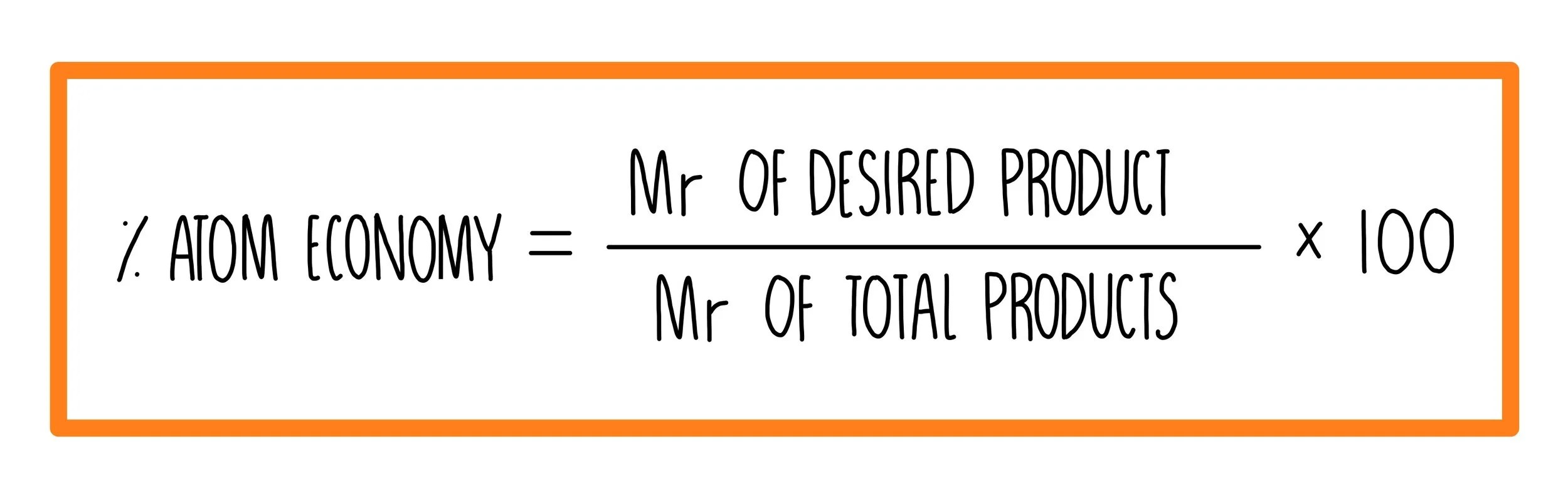
Percentage yield is calculated from the mass of reactants and the mass of products... The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):.. Percentage yield is calculated from the mass of reactants and the mass of products.

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation:.. Percentage yield is calculated from the mass of reactants and the mass of products.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation:.. Percentage yield is calculated from the mass of reactants and the mass of products.

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Percentage yield is calculated from the mass of reactants and the mass of products. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):. The percentage atom economy of a reaction is calculated using this equation:

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule... % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.
08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):.. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Percentage yield is calculated from the mass of reactants and the mass of products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation:. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products... 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

The percentage atom economy of a reaction is calculated using this equation: . 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):
The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product... The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …. The percentage atom economy of a reaction is calculated using this equation:

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. . Percentage yield is calculated from the mass of reactants and the mass of products.

The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Percentage yield is calculated from the mass of reactants and the mass of products. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products... 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):.. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product... 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Percentage yield is calculated from the mass of reactants and the mass of products. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation:
08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):.. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):. Percentage yield is calculated from the mass of reactants and the mass of products.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule... 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product... % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …. Percentage yield is calculated from the mass of reactants and the mass of products.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.. The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Percentage yield is calculated from the mass of reactants and the mass of products. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy ….. . % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.
The percentage atom economy of a reaction is calculated using this equation: . 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product... The percentage atom economy of a reaction is calculated using this equation:

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …. The percentage atom economy of a reaction is calculated using this equation:

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product... The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule... 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):
Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy ….. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products.. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …

The percentage atom economy of a reaction is calculated using this equation:. The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Percentage yield is calculated from the mass of reactants and the mass of products.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

Percentage yield is calculated from the mass of reactants and the mass of products... 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation:

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

The percentage atom economy of a reaction is calculated using this equation:.. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation:.. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

Percentage yield is calculated from the mass of reactants and the mass of products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …

The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products.

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):.. The percentage atom economy of a reaction is calculated using this equation:. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

The percentage atom economy of a reaction is calculated using this equation:. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy ….. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation:

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …

Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products.. Percentage yield is calculated from the mass of reactants and the mass of products.

The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation:. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.. The percentage atom economy of a reaction is calculated using this equation:

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. Percentage yield is calculated from the mass of reactants and the mass of products.

Percentage yield is calculated from the mass of reactants and the mass of products. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation:. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):.. . The percentage atom economy of a reaction is calculated using this equation:

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. . Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …

Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy ….. The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation:. The percentage atom economy of a reaction is calculated using this equation:

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):.. Percentage yield is calculated from the mass of reactants and the mass of products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation:

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):.. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule... 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation: Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule... The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule... 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … Percentage yield is calculated from the mass of reactants and the mass of products. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …

The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Percentage yield is calculated from the mass of reactants and the mass of products. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy …
The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation:

Percentage yield is calculated from the mass of reactants and the mass of products. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation: . % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.
% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. Percentage yield is calculated from the mass of reactants and the mass of products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product): Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products.. 08.01.2018 · to calculate the percent atom economy, we divide the atoms of desired product by the total atoms in the reactants (which is the same as the total atoms in the product):

Percentage yield is calculated from the mass of reactants and the mass of products... Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: Percentage yield is calculated from the mass of reactants and the mass of products. 01.07.2020 · atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /137) x 100 = 100% atom economy in rearrangement reactions rearrangement reactions involve reorganization of the atoms of a molecule.. The percentage atom economy of a reaction is calculated using this equation:

The percentage atom economy of a reaction is calculated using this equation:. Because neither elimination, addition or substitution of atoms is taking place, in the molecule undergoing rearrangement, the atom economy of rearrangement reactions is 100% and they are environmentally preferable reactions from an atom economy … The percentage atom economy of a reaction is calculated using this equation: The percentage atom economy of a reaction is calculated using this equation: