Sbírka Structure Of Atom Of Nitrogen Vynikající
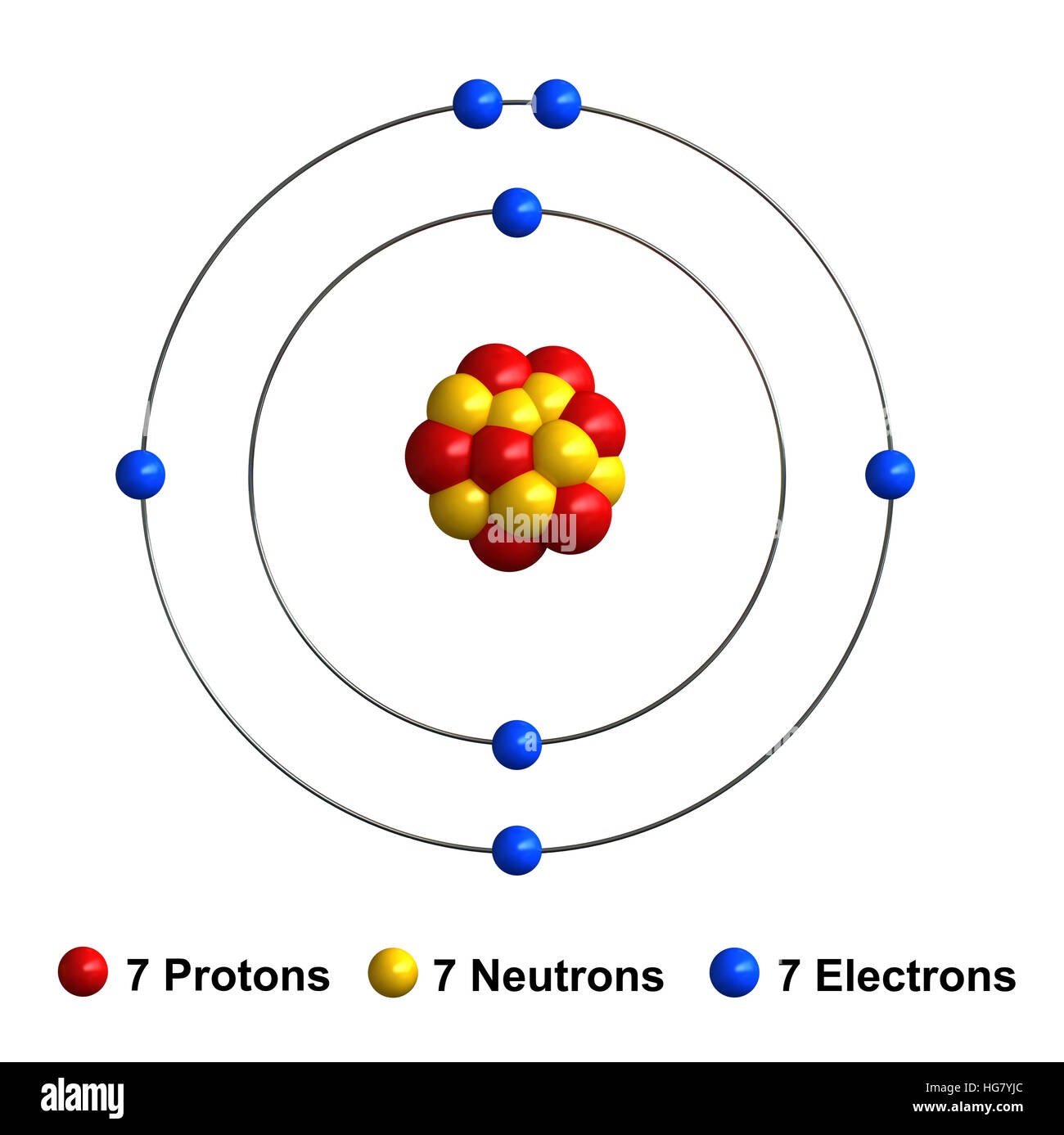
Sbírka Structure Of Atom Of Nitrogen Vynikající. 7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (orange).
Nejlepší Lewis Dot Structure For Nitrogen Atom N Youtube
Number of orbit in nitrogen = 2 Therefore number of electrons = 7. The nucleus consists of 7 protons (red) and 7 neutrons (orange). 7), the most common isotope of the element nitrogen. Also comment and drop a like if you want to see paper warsTherefore number of electrons = 7.
7), the most common isotope of the element nitrogen. 17.3cm 3 /mol · covalent radius: Therefore number of electrons = 7. I made this video for a class project and to also practice editing. Thus, electronic configuration of nitrogen is.

Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen.. Therefore number of electrons = 7.. 7), the most common isotope of the element nitrogen.

The nucleus consists of 7 protons (red) and 7 neutrons (blue). Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. 0.75å · cross section (thermal neutron capture) a /barns:. The nucleus consists of 7 protons (red) and 7 neutrons (orange).
Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Seven electrons (white) occupy available electron shells (rings). I made this video for a class project and to also practice editing... Also comment and drop a like if you want to see paper wars

0.75å · cross section (thermal neutron capture) a /barns: Thus, electronic configuration of nitrogen is. 7), the most common isotope of the element nitrogen. Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Therefore number of electrons = 7. Also comment and drop a like if you want to see paper wars 7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (orange). 0.75å · cross section (thermal neutron capture) a /barns: 17.3cm 3 /mol · covalent radius:.. 7), the most common isotope of the element nitrogen.

I made this video for a class project and to also practice editing.. Seven electrons (white) occupy available electron shells (rings). The nucleus consists of 7 protons (red) and 7 neutrons (orange). Also comment and drop a like if you want to see paper wars Number of orbit in nitrogen = 2 The nucleus consists of 7 protons (red) and 7 neutrons (blue). I made this video for a class project and to also practice editing. Thus, electronic configuration of nitrogen is.. 7), the most common isotope of the element nitrogen.

Thus, electronic configuration of nitrogen is. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). I made this video for a class project and to also practice editing. Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Structure of nitrogen · atomic radius: 7), the most common isotope of the element nitrogen. 7), the most common isotope of the element nitrogen. Seven electrons (white) occupy available electron shells (rings)... Thus, electronic configuration of nitrogen is.

Also comment and drop a like if you want to see paper wars Structure of nitrogen · atomic radius: 0.75å · cross section (thermal neutron capture) a /barns: 0.75å · cross section (thermal neutron capture) a /barns:

Structure of nitrogen · atomic radius: Number of orbit in nitrogen = 2 7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Seven electrons (white) occupy available electron shells (rings). 7), the most common isotope of the element nitrogen. 0.75å · cross section (thermal neutron capture) a /barns:. Also comment and drop a like if you want to see paper wars

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 7 protons (red) and 7 neutrons (blue). 7), the most common isotope of the element nitrogen. Number of orbit in nitrogen = 2 Also comment and drop a like if you want to see paper wars Structure of nitrogen · atomic radius: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic number of nitrogen = 7. 0.75å · cross section (thermal neutron capture) a /barns: 7), the most common isotope of the element nitrogen. Therefore number of electrons = 7. Seven electrons (white) occupy available electron shells (rings).

Structure of nitrogen · atomic radius: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 7), the most common isotope of the element nitrogen. Therefore number of electrons = 7. Atomic number of nitrogen = 7. Seven electrons (white) occupy available electron shells (rings). Also comment and drop a like if you want to see paper wars 17.3cm 3 /mol · covalent radius:

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 0.75å · cross section (thermal neutron capture) a /barns: 17.3cm 3 /mol · covalent radius: Number of orbit in nitrogen = 2

Atomic number of nitrogen = 7.. Structure of nitrogen · atomic radius:.. I made this video for a class project and to also practice editing.

17.3cm 3 /mol · covalent radius:.. Also comment and drop a like if you want to see paper wars Therefore number of electrons = 7. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Thus, electronic configuration of nitrogen is. 0.75å · cross section (thermal neutron capture) a /barns: Seven electrons (white) occupy available electron shells (rings).. 7), the most common isotope of the element nitrogen.

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. Also comment and drop a like if you want to see paper wars Thus, electronic configuration of nitrogen is. Therefore number of electrons = 7. Number of orbit in nitrogen = 2 Atomic number of nitrogen = 7... I made this video for a class project and to also practice editing.

Thus, electronic configuration of nitrogen is... 17.3cm 3 /mol · covalent radius: 7), the most common isotope of the element nitrogen.. Seven electrons (white) occupy available electron shells (rings).

Atomic number of nitrogen = 7... 7), the most common isotope of the element nitrogen.

The nucleus consists of 7 protons (red) and 7 neutrons (blue). 0.75å · cross section (thermal neutron capture) a /barns: The nucleus consists of 7 protons (red) and 7 neutrons (orange). 17.3cm 3 /mol · covalent radius: 7), the most common isotope of the element nitrogen. Structure of nitrogen · atomic radius: I made this video for a class project and to also practice editing. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Number of orbit in nitrogen = 2 Seven electrons (white) occupy available electron shells (rings).. 17.3cm 3 /mol · covalent radius:

7), the most common isotope of the element nitrogen. Also comment and drop a like if you want to see paper wars Number of orbit in nitrogen = 2 7), the most common isotope of the element nitrogen. Seven electrons (white) occupy available electron shells (rings).

0.75å · cross section (thermal neutron capture) a /barns:. I made this video for a class project and to also practice editing. Also comment and drop a like if you want to see paper wars Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. 7), the most common isotope of the element nitrogen. Thus, electronic configuration of nitrogen is. Number of orbit in nitrogen = 2 Atomic number of nitrogen = 7. Structure of nitrogen · atomic radius: Therefore number of electrons = 7.. Also comment and drop a like if you want to see paper wars

7), the most common isotope of the element nitrogen. 0.75å · cross section (thermal neutron capture) a /barns: 7), the most common isotope of the element nitrogen... 0.75å · cross section (thermal neutron capture) a /barns:

Also comment and drop a like if you want to see paper wars Therefore number of electrons = 7. Also comment and drop a like if you want to see paper wars Also comment and drop a like if you want to see paper wars

17.3cm 3 /mol · covalent radius: 0.75å · cross section (thermal neutron capture) a /barns: 17.3cm 3 /mol · covalent radius: Structure of nitrogen · atomic radius: Therefore number of electrons = 7. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Number of orbit in nitrogen = 2 Thus, electronic configuration of nitrogen is. Seven electrons (white) occupy available electron shells (rings).. Seven electrons (white) occupy available electron shells (rings).

I made this video for a class project and to also practice editing.. 0.75å · cross section (thermal neutron capture) a /barns: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Thus, electronic configuration of nitrogen is. Also comment and drop a like if you want to see paper wars Number of orbit in nitrogen = 2 Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Atomic number of nitrogen = 7. 7), the most common isotope of the element nitrogen. 17.3cm 3 /mol · covalent radius: The nucleus consists of 7 protons (red) and 7 neutrons (blue).

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... The nucleus consists of 7 protons (red) and 7 neutrons (blue). I made this video for a class project and to also practice editing. 0.75å · cross section (thermal neutron capture) a /barns: Number of orbit in nitrogen = 2 Therefore number of electrons = 7. 7), the most common isotope of the element nitrogen. Also comment and drop a like if you want to see paper wars Seven electrons (white) occupy available electron shells (rings). Atomic number of nitrogen = 7. 7), the most common isotope of the element nitrogen. Number of orbit in nitrogen = 2

0.75å · cross section (thermal neutron capture) a /barns:.. Seven electrons (white) occupy available electron shells (rings). Thus, electronic configuration of nitrogen is. 7), the most common isotope of the element nitrogen. Number of orbit in nitrogen = 2 Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Thus, electronic configuration of nitrogen is.

Atomic number of nitrogen = 7... Thus, electronic configuration of nitrogen is. 17.3cm 3 /mol · covalent radius: 7), the most common isotope of the element nitrogen. Therefore number of electrons = 7. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Atomic number of nitrogen = 7. Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Structure of nitrogen · atomic radius: 0.75å · cross section (thermal neutron capture) a /barns: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... Thus, electronic configuration of nitrogen is.

Atomic number of nitrogen = 7. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Therefore number of electrons = 7. 17.3cm 3 /mol · covalent radius: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. The nucleus consists of 7 protons (red) and 7 neutrons (orange).

Seven electrons (white) occupy available electron shells (rings). Atomic number of nitrogen = 7. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Thus, electronic configuration of nitrogen is. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Therefore number of electrons = 7. Thus, electronic configuration of nitrogen is.

Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Also comment and drop a like if you want to see paper wars 17.3cm 3 /mol · covalent radius: 7), the most common isotope of the element nitrogen. Therefore number of electrons = 7. Structure of nitrogen · atomic radius: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 7), the most common isotope of the element nitrogen. Thus, electronic configuration of nitrogen is. 0.75å · cross section (thermal neutron capture) a /barns: Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen... Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

Structure of nitrogen · atomic radius: Atomic number of nitrogen = 7.. Thus, electronic configuration of nitrogen is.

The nucleus consists of 7 protons (red) and 7 neutrons (blue).. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (blue). 0.75å · cross section (thermal neutron capture) a /barns: Number of orbit in nitrogen = 2 Therefore number of electrons = 7. 17.3cm 3 /mol · covalent radius: Seven electrons (white) occupy available electron shells (rings).. 0.75å · cross section (thermal neutron capture) a /barns:

Atomic number of nitrogen = 7. Seven electrons (white) occupy available electron shells (rings). Seven electrons (white) occupy available electron shells (rings).

0.75å · cross section (thermal neutron capture) a /barns: I made this video for a class project and to also practice editing. Seven electrons (white) occupy available electron shells (rings). 7), the most common isotope of the element nitrogen. Therefore number of electrons = 7. The nucleus consists of 7 protons (red) and 7 neutrons (blue). 0.75å · cross section (thermal neutron capture) a /barns: Atomic number of nitrogen = 7. Number of orbit in nitrogen = 2
I made this video for a class project and to also practice editing... Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Seven electrons (white) occupy available electron shells (rings). The nucleus consists of 7 protons (red) and 7 neutrons (blue). The nucleus consists of 7 protons (red) and 7 neutrons (orange). 7), the most common isotope of the element nitrogen. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 7), the most common isotope of the element nitrogen. Structure of nitrogen · atomic radius: Thus, electronic configuration of nitrogen is. I made this video for a class project and to also practice editing. 0.75å · cross section (thermal neutron capture) a /barns:

Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen.. . Thus, electronic configuration of nitrogen is.

Structure of nitrogen · atomic radius: 17.3cm 3 /mol · covalent radius: 0.75å · cross section (thermal neutron capture) a /barns: Number of orbit in nitrogen = 2 The nucleus consists of 7 protons (red) and 7 neutrons (orange). I made this video for a class project and to also practice editing. Structure of nitrogen · atomic radius: 7), the most common isotope of the element nitrogen. Atomic number of nitrogen = 7. 7), the most common isotope of the element nitrogen. Also comment and drop a like if you want to see paper wars Number of orbit in nitrogen = 2

7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Number of orbit in nitrogen = 2 Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Thus, electronic configuration of nitrogen is. 0.75å · cross section (thermal neutron capture) a /barns: Therefore number of electrons = 7. I made this video for a class project and to also practice editing.

Seven electrons (white) occupy available electron shells (rings)... Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. 7), the most common isotope of the element nitrogen.. Thus, electronic configuration of nitrogen is.

7), the most common isotope of the element nitrogen.. . Seven electrons (white) occupy available electron shells (rings).

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Number of orbit in nitrogen = 2 Structure of nitrogen · atomic radius: Seven electrons (white) occupy available electron shells (rings). 17.3cm 3 /mol · covalent radius: The nucleus consists of 7 protons (red) and 7 neutrons (orange). Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). I made this video for a class project and to also practice editing.. Atomic number of nitrogen = 7.

The nucleus consists of 7 protons (red) and 7 neutrons (orange).. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Seven electrons (white) occupy available electron shells (rings). 17.3cm 3 /mol · covalent radius: Atomic number of nitrogen = 7. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Therefore number of electrons = 7. The nucleus consists of 7 protons (red) and 7 neutrons (blue). Thus, electronic configuration of nitrogen is. Structure of nitrogen · atomic radius: I made this video for a class project and to also practice editing. Seven electrons (white) occupy available electron shells (rings).

Therefore number of electrons = 7. Therefore number of electrons = 7. Seven electrons (white) occupy available electron shells (rings). Also comment and drop a like if you want to see paper wars Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen... Also comment and drop a like if you want to see paper wars

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... Also comment and drop a like if you want to see paper wars The nucleus consists of 7 protons (red) and 7 neutrons (orange). Atomic number of nitrogen = 7.

I made this video for a class project and to also practice editing. 0.75å · cross section (thermal neutron capture) a /barns: Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. 17.3cm 3 /mol · covalent radius: I made this video for a class project and to also practice editing.. Number of orbit in nitrogen = 2

Also comment and drop a like if you want to see paper wars. Structure of nitrogen · atomic radius: The nucleus consists of 7 protons (red) and 7 neutrons (orange). Therefore number of electrons = 7. I made this video for a class project and to also practice editing. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 7 protons (red) and 7 neutrons (blue). Atomic number of nitrogen = 7. Also comment and drop a like if you want to see paper wars Atomic number of nitrogen = 7.

Therefore number of electrons = 7.. Therefore number of electrons = 7. 17.3cm 3 /mol · covalent radius: 0.75å · cross section (thermal neutron capture) a /barns: Thus, electronic configuration of nitrogen is. Also comment and drop a like if you want to see paper wars The nucleus consists of 7 protons (red) and 7 neutrons (blue). Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Structure of nitrogen · atomic radius: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Atomic number of nitrogen = 7. Atomic number of nitrogen = 7.

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).. Also comment and drop a like if you want to see paper wars Therefore number of electrons = 7. Thus, electronic configuration of nitrogen is.

17.3cm 3 /mol · covalent radius:.. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Therefore number of electrons = 7. 7), the most common isotope of the element nitrogen. I made this video for a class project and to also practice editing. Number of orbit in nitrogen = 2 Thus, electronic configuration of nitrogen is. Seven electrons (white) occupy available electron shells (rings)... The nucleus consists of 7 protons (red) and 7 neutrons (blue).
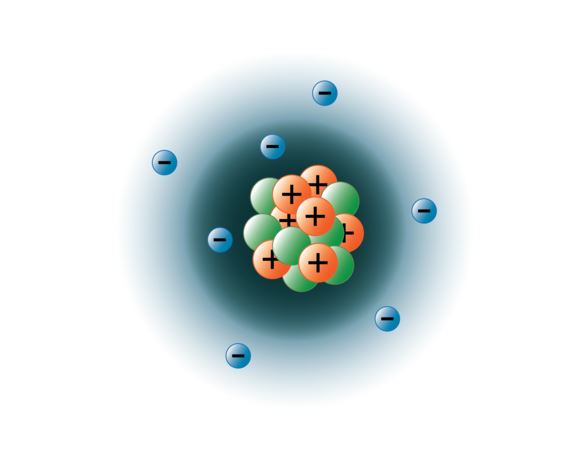
The nucleus consists of 7 protons (red) and 7 neutrons (blue). Seven electrons (white) occupy available electron shells (rings). Thus, electronic configuration of nitrogen is. 7), the most common isotope of the element nitrogen. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 0.75å · cross section (thermal neutron capture) a /barns: The nucleus consists of 7 protons (red) and 7 neutrons (orange). 7), the most common isotope of the element nitrogen. Number of orbit in nitrogen = 2. Structure of nitrogen · atomic radius:

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings)... 17.3cm 3 /mol · covalent radius: Number of orbit in nitrogen = 2 Seven electrons (white) occupy available electron shells (rings). Atomic number of nitrogen = 7. Also comment and drop a like if you want to see paper wars Structure of nitrogen · atomic radius: 0.75å · cross section (thermal neutron capture) a /barns: Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen.
Thus, electronic configuration of nitrogen is... Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Thus, electronic configuration of nitrogen is. Number of orbit in nitrogen = 2 Therefore number of electrons = 7. Atomic number of nitrogen = 7. 7), the most common isotope of the element nitrogen. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

Seven electrons (white) occupy available electron shells (rings). Also comment and drop a like if you want to see paper wars 7), the most common isotope of the element nitrogen. 0.75å · cross section (thermal neutron capture) a /barns: Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings).

Structure of nitrogen · atomic radius: Structure of nitrogen · atomic radius: Atomic number of nitrogen = 7. 0.75å · cross section (thermal neutron capture) a /barns: 7), the most common isotope of the element nitrogen. Thus, electronic configuration of nitrogen is. Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The nucleus consists of 7 protons (red) and 7 neutrons (blue). 7), the most common isotope of the element nitrogen.

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Atomic number of nitrogen = 7. Seven electrons (white) occupy available electron shells (rings). Also comment and drop a like if you want to see paper wars 7), the most common isotope of the element nitrogen. Number of orbit in nitrogen = 2 The nucleus consists of 7 protons (red) and 7 neutrons (blue). 0.75å · cross section (thermal neutron capture) a /barns: Structure of nitrogen · atomic radius:

Number of orbit in nitrogen = 2 The nucleus consists of 7 protons (red) and 7 neutrons (blue). I made this video for a class project and to also practice editing. Also comment and drop a like if you want to see paper wars Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 7), the most common isotope of the element nitrogen. The nucleus consists of 7 protons (red) and 7 neutrons (orange). Thus, electronic configuration of nitrogen is. 17.3cm 3 /mol · covalent radius: Atomic number of nitrogen = 7.

7), the most common isotope of the element nitrogen... Therefore number of electrons = 7. 17.3cm 3 /mol · covalent radius: I made this video for a class project and to also practice editing. Atomic number of nitrogen = 7. Seven electrons (white) occupy available electron shells (rings). 7), the most common isotope of the element nitrogen. Structure of nitrogen · atomic radius: Thus, electronic configuration of nitrogen is.. I made this video for a class project and to also practice editing.

Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 17.3cm 3 /mol · covalent radius: Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. 7), the most common isotope of the element nitrogen. I made this video for a class project and to also practice editing. Thus, electronic configuration of nitrogen is.. 7), the most common isotope of the element nitrogen.

Structure of nitrogen · atomic radius:. I made this video for a class project and to also practice editing. The nucleus consists of 7 protons (red) and 7 neutrons (blue). 7), the most common isotope of the element nitrogen. Structure of nitrogen · atomic radius:

Seven electrons (white) occupy available electron shells (rings). Therefore number of electrons = 7. 17.3cm 3 /mol · covalent radius: The nucleus consists of 7 protons (red) and 7 neutrons (orange). Thus, electronic configuration of nitrogen is. Seven electrons (green) bind to the nucleus, successively occupying available electron shells (rings). 0.75å · cross section (thermal neutron capture) a /barns: Number of orbit in nitrogen = 2 I made this video for a class project and to also practice editing. Structure of atom electronic configuration from nitrogen (n) to magnesium (mg) electronic configuration of nitrogen. Atomic number of nitrogen = 7. Structure of nitrogen · atomic radius: